Pipeline of GD & GBA-PD
Venglustat (Ibiglustat)
Clinical Trials Of Venglustat
| phase | study | target pt | status / timing | number | design | Tx | primary outcome | secondary / result notes | trial ID |
|---|---|---|---|---|---|---|---|---|---|
| P2 | LEAP |
GD type 1&3 (+ cerezyme), Adult Start 2017 Jan. Completion: May 2024 (>7년!) | recruiting | 10 | open-label, single-group | Venglustat (15 mg/day); 52w |
Part 1: GD 1&3 -> Part 2: GD3 age ≥18 with horizontal saccade eye movement (HSEM) impairment, received ERT for ≥3 years with a stable imiglucerase dose for ≥6 months before enrollment, and achieved therapeutic goals, No Hx of splenectomy, No myoclonic seizure. |
Keep in mind that they don't look at GBA activity due to its mechanism. Secondary: efficacy in neurological function (mSST) and exploratory CSF biomarkers; efficacy in interstitial lung disease. 4 of 5 patients exhibited regression of ILD on high-resolution CT. 2 patients experienced spleen-volume reduction ≥10%. Platelet count increased by 17% (range -13% to 42%) from baseline to Week 26. The 3 patients with lowest baseline platelet counts had increases of 23-42%. Neurological manifestations (mSST) showed no deterioration from baseline; saccade only measured horizontal. | NCT02843035 |
| P2 | MOVES-PD, POC study |
GBA+PD (Heteroz) Start 2016 Dec. Primary Completion (LPLV일걸): 2020 Dec. Data read-out from this study is expected in 2021 H1; study completion February 2023. Early PD, heterozygous carriers. Patients carrying known sequence variants associated with GBA-PD, such as E326K, must have rapid eye movement (REM) sleep behavior disorder (RBD) confirmed by historically documented polysomnography or by questionnaire. Has symptoms of PD ≥2 years. H and Y stage of 2 or lower at baseline. Venglustat (15 mg/day) or matching placebo. | Recruiting, read-out in H1 2021 -> stop 20210205 |
243 Part 2: venglustat군 108명, 위약 약 108명 |
Placebo controlled Part 1: 4 groups (n of 4-5 each group) Part 1: 4-week dose escalation study at 3 different doses in a sequential cohort design; maximum follow-up of 36 weeks. -> Part 2: "single dose" 52w (followed by a 156-week extension period) |
Part 1: 66w Part 2: 170w | Primary outcome: MDS-UPDRS II & III |
Secondary outcome measures: Parkinson's Disease Cognitive Rating Scale, MDS-UPDRS I, II, & III, H&Y score. Included patients: N370S (52.8%), L444P (18.4%), E326K (16.6%), T369 (3.7%). Mean MDS-UPDRS II+III: 36.9 (16.7). Mean MoCA: 27.9 (2.4). AGE: 59 Y. Disease duration: 6.6 (4.3). 패인: 너무 mild GBA mutation (N370S가 반) 위주였나? | NCT02906020 |
LEAP Biomarker Notes
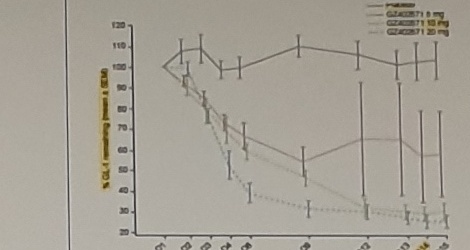
low plasma GlcCer (js it takes about 1 w to get plateau)
2021 WORLDSymposium, LLOQ
| PLASMA | CSF | |
|---|---|---|
| GlcCer | 0.1 ug/ml | 2.0 ng/ml |
| GlcSph | 5.0 ng/ml | 5 pg/ml |
change from baseline at 52w (Schiffmann, 2023 #2578)
| PLASMA | CSF | note | |
|---|---|---|---|
| GlcCer | ↓78% (46-84) | ↓81% (47-83) | 모두 w4부터 크게 drop, but CSF GlcSph?! |
| GlcSph | ↓56% (23-60) | ↓70% (45-76) | 점진적 w4 ↓31.7% -> w26 ↓47.8 |
normal control 군 없음 유념! Js: 이건 GCSi라서 GlcCer보다 GlcSph가 더 천천히, & 적게 주는 듯함.
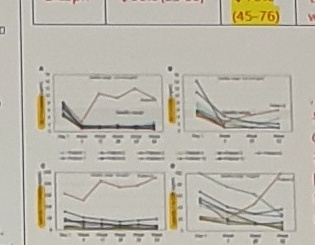
[SARA]
Baseline: 대개 3정도인듯
(2020 world)
Trail making test: All patients remained stable or improved
Brain vol (2021worlds): ↑ regional vol, ↓ ventricle vol, =whole brain vol
fMRI: Improved, ie connectivity
no changes in horizontal saccadic eye movement from baseline
Results: (Schiffmann 2023) SARA improved.
MOVES-PD Biomarker And Clinical Results
[part 1 결과] GlcCer reduction; 79% in plasma and 74% in CSF.
(normal control 불참여이므로 GlcCer가 normal control 대비 원래 증가되어 있는지는 불명.)
Plasma biomarkers (at 2 & 4w):
- All patients with available data had GlcCer levels above the lower limit of quantification (LLOQ) from baseline until Week 4.
- All patients had plasma GlcSph levels below the LLOQ from baseline to Week 4.
CSF biomarkers (at 4ws):
- All patients at baseline had GL-1 levels above the LLOQ.
- Most patients had CSF lyso-GL-1 levels below the LLOQ at baseline and Week 4.
202103 AD/PD:
At week 52, least-squares mean ± SE MDS-UPDRS Part II+III score changes from baseline were 7.29 ± 1.36 (venglustat) and 4.71 ± 1.27 (placebo; P=NS for between-group difference). Most adverse events (AEs) were mild/moderate; serious AEs were reported in 10.9% and 10.8% of participants in the venglustat and placebo groups, respectively.
[part 2 결과] GlcCer reduction; 75% in plasma and CSF.
Results: (Giladi, 2023 #2529)
NfL concentrations in CSF were elevated by approximately 20% versus baseline in participants in the venglustat group (non-significant w52 vs BL), although they remained stable in the placebo group. No between-group differences in plasma NfL concentrations were evident (appendix pp 17, 29). No notable differences were observed between the venglustat and placebo groups for any other plasma or CSF biomarkers assessed. DaTSCAN showed a reduction in dopamine transporter density in both groups; no discernible between-group differences were observed in any brain region (appendix p 18). CSF total aSyn: no change venglustat vs placebo.
Prevail PR001
Preclinical
| section | model | route / dose | visible notes |
|---|---|---|---|
| ? | In vitro |
No PR001: vehicle only; Low dose: 2 × 10^5 vg/cell PR001; High dose: 2 × 10^6 vg/cell PR001. No PR001: vehicle only; Low dose: 1.3 × 10^5 vg/cell PR001; High dose: 1.3 × 10^6 vg/cell PR001. | Each bar represents the mean ± SEM. |
| GD | CBE mice (단기투여) |
intracerebroventricular The low dose was 3.2 × 10^9 vg (vector genome); the medium dose was 1 × 10^10 vg; the high dose was 3.2 × 10^10 vg. | CBE-treated mice exhibited reduced performance on tests of motor function (rotarod and tapered beam). The highest dose of PR001 resulted in a statistically significant improvement in motor function. GCase activity in the brain was positively correlated with performance on the rotarod test and negatively correlated with glycolipid levels in the brain in a statistically significant manner. |

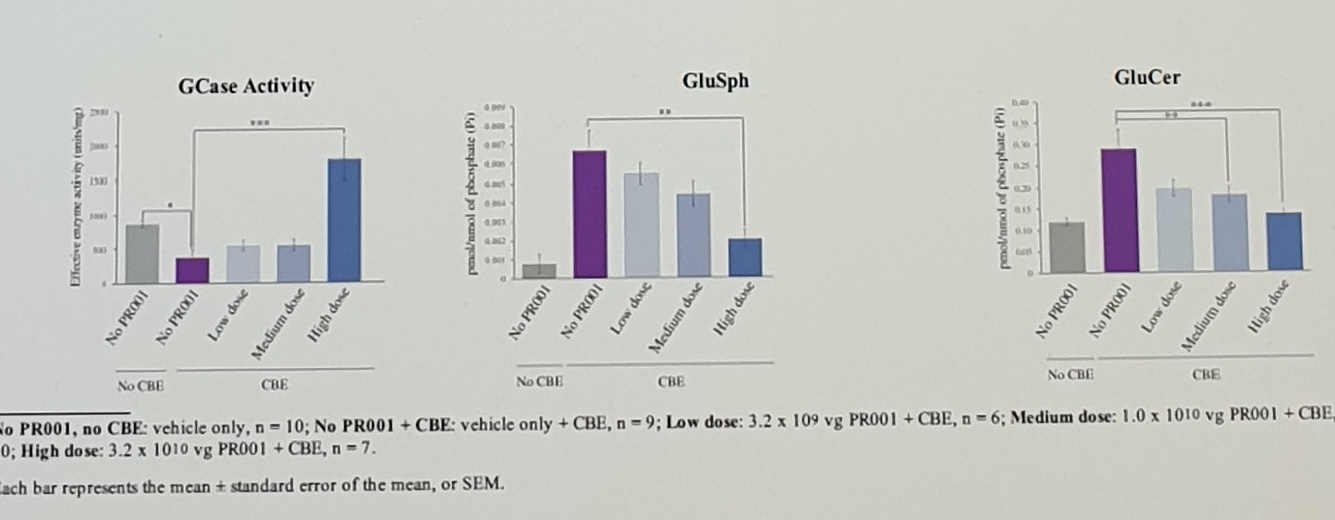
No PR001, no CBE: vehicle only, n = 10; No PR001 + CBE: vehicle only + CBE, n = 9; Low dose: 3.2 × 10^9 vg PR001 + CBE, n = 6; Medium dose: 1.0 × 10^10 vg PR001 + CBE, n = 10; High dose: 3.2 × 10^10 vg PR001 + CBE, n = 7.
Each bar represents the mean ± standard error of the mean, or SEM.
Astrogliosis
Glial Scarring
Microgliosis
Iba1 Immunoreactivity
Uncertain Spans
| location | unresolved text | reason |
|---|---|---|
| LEAP row | GD type 1&3 (+ cerezyme), Adult; Part 1: GD 1&3 -> Part 2: GD3 | Cell is split by grid lines; current transcription preserves the visible wording. |
| LEAP eligibility | HSEM / ERT / stable imiglucerase sentence | Small text, but OCR and visual reading agree broadly. |
| LEAP biomarker values | ↓81% (47-83), ↓70% (45-76), w4 ↓31.7% -> w26 ↓47.8 | High-risk numeric transcription from dense highlighted cells. |
| MOVES-PD design | Part 1: 66w, Part 2: 170w | Duration cells coexist with detailed design text; row alignment may need adjacent-photo interpretation. |
| MOVES-PD baseline | Disease duration: 6.6 (4.3) | Parenthetical value is small. |
| MOVES-PD mutation list | T369 (3.7%) | Mutation shorthand is small. |
| PR001 in vitro captions | 2 × 10^5, 2 × 10^6, 1.3 × 10^5, 1.3 × 10^6 vg/cell | Plot captions are small; images are retained. |
| PR001 lower plots | Astrogliosis / Microgliosis plots | Lower plots are cut off and continue in 20240722_181805. |