Pipeline of GD & GBA-PD
Ambroxol
Clinical Trials Of Ambroxol
| phase / program | sponsor / ID | target / eligibility | number / design | Tx | primary outcome | secondary / result notes |
|---|---|---|---|---|---|---|
| P2, AMBITIOUS |
Italy NCT05287503 EudraCT 2021-004565-13 |
patients with baseline GBA activity not very low Inclusion: Duration of motor symptoms >5 years Exclusion: 2. PDD (MDS Level II criteria) 3. Hoehn & Yahr stage ≥ 4/5 in the medication-ON condition 4. DBS | 52-week treatment period | Primary: i) MoCA change, ii) Rate of conversion from normal cognitive status to MCI, or from MCI to overt dementia over the 52-week treatment period. |
MDS-UPDRS II, III GBA activity (CSF, WBC), csf bm of neurodegeneration, functional connectivity at rest with brain MRI (Colucci 2023) Sample size calculation: The primary endpoint is the change in total MoCA score. Considering (1) an average decrease in 24 months in the total MoCA score of 1±2 points in the general PD population (0.5±2 points/year), (Colucci 2023) (2) a more rapid decline of MoCA score even in asymptomatic carriers of GBA gene mutations compared with non-carriers, 58 (3) expecting a threefold greater risk for dementia in GBA-PD than non-carriers underlain by a with faster rate of MoCA score decrease (3±3 points in 24 months) with a linear pattern of decline-13 and (4) assuming that ABX treatment will reduce this increase by 50% (1.5±2 points in 24 months), the sample size sufficient to have 80% power with a type I error of 20% is 54 patients (27 ABX, 27 placebo). Considering an expected dropout rate of 10%, it is necessary to enrol at least 60 patients (30 in each arm). | |
| P3, ASPro-PD | UCL, NCT05778617 |
GPA-PD: ambroxol vs placebo, 52w, 65명 1200 mg (400 mg TID) | RCT, DB, placebo | 52w | N=330, PLACEBO-CONTROLLED, TX DURATION 2Y; PRIMARY: mds-updrs I+II+III | |
| P2, AiM-PD | UCL; NCT02941822 |
PD (20 patients, 10 GBA-positive & 10 GBA-negative status) diagnosed with PD 1-III GBA+PD: 8명 (p.E326K wild type (3 patients), p.N370S wild type (1 patient), p.R463C* wild type (2 patients), p.T369M/p.W393X* (1 patient), and RecNcil (p.L444P, p.A456P, and p.V460V)* wild type (1 patient)) GBA-PD: 10명 | Single group, OL, No placebo | 6 m | Primary: GBA activity & ambroxol levels in blood & CSF |
Results: (Mullin et al. 2020, PMID 31930374) fig2c) CSF GBA protein: BL에서 GBA+PD (black dot)가 GBA-PD보다 꽤 낮은 경향, 6m 후 ↑135% (두 군 모두 증가로 보임). fig2a) CSF GBA activity: BL에서 GBA+PD (black dot)가 GBA-PD보다 뚜렷이 낮았는데, 6m 후 ↓19% (두 군 모두 감소로 보임). CSF에 있는 ambroxol 때문에 감소한 것으로 해석. Ambroxol is a pH-dependent inhibitory small molecular chaperone that binds to the active site of the GCase protein and reduces activity. Binding enables transportation to the lysosome and elution of free active enzymes under acidic conditions. Therefore, in acellular CSF, ambroxol will bind to and inhibit free GCase. WBC GBA activity: BL에서 GBA+PD가 GBA-PD보다 뚜렷이 낮았는데, 치료 직후 두 군 모두 ↑ -> 이후 다시 내려가서 6m 후엔 BL과 유사수준됨. CSF T-a-syn: BL에서 GBA+PD가 GBA-PD보다 좀 낮은듯 보이기도, 6m 후 ↑13% (두 군 모두). 해석: d/t an increase of extracellular export of the a-syn from the brain parenchyma. MDS-UPDRS III: ↓ by 6.8 (7.1)p (statistically significant). |
Adult GD is responsive too (narita 2016)
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| GD type | 3 | 3 | 3 | 2 | 3 |
| Age | 28 | 20 | 15 | 3 | 25 |
우리 거: GD: Pts at baseline: adult, myoclonus (responsive), GBA activity should not be not possible.
GlcSph normal range: in dried blood spots ref, <4.8 ng/mL (Kim, 2020 #809), in CSF <10.0 pg/mL (from n=37 control) (Narita 2016)
Venglustat (Ibiglustat)
Preclinical
2017 Sardi www.pnas.org/cgi/doi/10.1073/pnas.1616152114 (이 논문에서 Venglustat 의 용량은 not-disclosed)
| row | GD Model | CBE model | PD only model |
|---|---|---|---|
| model / source | GbaD409V/D409V, (Sardi et al. 2017, PMID) | CBE (Marshall et al. 2016, PMID 26948439) | A53T-SNCA mouse (without mutations in Gba); (Sardi et al. 2017, PMID), venglustat자체는 아니고 closely related compound (GZ667161). |
| Tx | 7m (age 6m-13m) | 6.5m (age 1.5m-8m), 30 mg/kg/day | |
| GBA activity | 이 TG mice has only 20% residual activity -> venglustat had no effect (gba activator가 아니므로 당연) | gba activator가 아니므로 볼 필요 없음. | |
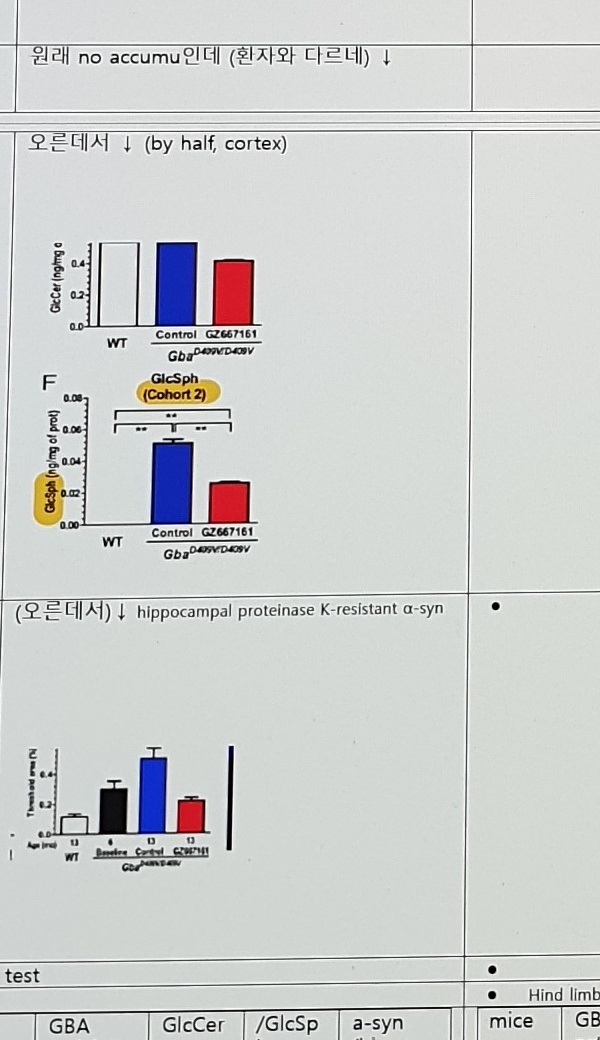
| GlcCer | 원래 no accumu인데 (환자와 다르네) ↓ | 원래 no accumu (despite reduced GBA activity by 20%, 이건 환자와 같네) 그렇지만 여기서 줄였음. fig4. | |
| GlcSph | 오른데서 ↓ (by half, cortex) | 원래 no accumu (despite reduced GBA activity by 20%), 원래 거의 적은 모양이니, 내릴 것도 없을 것이니 논문에 언급 없음. | |
| a-syn | (오른데서) ↓ hippocampal proteinase K-resistant α-syn |
No changes in the levels of cytosolic soluble α-syn. However, the levels of the membrane-associated and insoluble α-syn species were reduced. (WT는 논문에 Fig 5에 없는듯 한데 당연히 올라간 데서 줄인 것이겠지?) proteinase K-resistant α-syn 분석은 실패. | |
| Neuronal loss | test | Not tested | |
| Behavior | Hind limb splay | ↑ memory (novel object recognition test) |
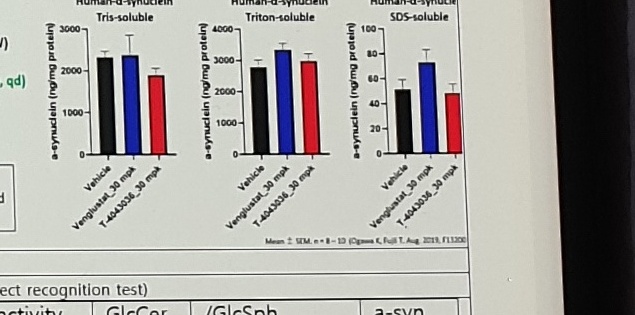
In House Data
Animal: A53T SNCA Tg mouse (from 7W)
Compound: Venglustat, T-4043036
Regimen: Repeated administration (po, qd)
30 mg/kg/day, po, 3 months
Tris-soluble: cytosolic
Triton-soluble: membrane-associated
SDS-soluble: insoluble
Summary Sub-Table: Venglustat Tx Effect
| model / row | GBA activity | GlcCer | GlcSph | a-syn |
|---|---|---|---|---|
| GbaD409V/D409V; dose / mice | Whole brain | Cortex | Cortex | na / hippo, insoluble |
| Tx effect Venglu | No effect (GCSi니까 당연) | ↓ (20%) | ↓ (50%) | ↓ to baseline (not revert) |
| CBE; Tx effect Venglu | (GCSi니까 무의미) | ↓ (>50%) | ↓ (50%) | na |
| A53T-SNCA; Tx effect Venglu | (GCSi니까 무의미) | ↓ (30%) | NA; No accumulation | ↓ (20%) |
Cathepsin D
Clinical Trials Of Venglustat
| phase | name | target pt | status | number | design | Tx | primary outcome | secondary | trial ID |
|---|---|---|---|---|---|---|---|---|---|
| P1 | HV | SAD | Safety | CSF is not mentioned. | NCT01674036 | ||||
| P1 | HV | 14 d | MAD, placebo | Safety |
PK CSF is not mentioned. [plasma] Glucosylceramide, Globotriaosylceramide (GL-3) and GM3 ganglioside CSF GlcSph: not detectible in healthy. | NCT01710826 |
Uncertain Spans
| location | unresolved text | reason |
|---|---|---|
| P2 AMBITIOUS row | EudraCT 2021-004565-13 | Split across multiple short lines; best visual reconstruction. |
| P3 ASPro-PD row | alignment of N=330, PLACEBO-CONTROLLED, TX DURATION 2Y | The right-side highlighted text may belong to this row or to an adjacent trial row. |
| AiM-PD genotype list | RecNcil (p.L444P, p.A456P, and p.V460V) | Small genotype shorthand; RecNcil may be an OCR/visual ambiguity. |
| adult GD note | GBA activity should not be not possible | Source wording is awkward and the line is low-resolution. |
| Venglustat model | GbaD409V/D409V | Subscript/superscript styling is flattened from the Word table. |
| Venglustat plots | exact y-axis values and significance marks | Embedded as image assets rather than guessed from low-resolution plots. |
| Clinical Trials of Venglustat | bottom rows after NCT01710826 | Rows after NCT01710826 are clipped by the photo’s lower edge. |