Pipeline of GD & GBA-PD
Eliglustat
| trial / phase | target patient | No | design | Tx | primary outcome | secondary / result notes | trial ID / citation |
|---|---|---|---|---|---|---|---|
| P3 EDGE | QD vs BID of eliglustat in patients with GD1 who have demonstrated clinical stability on eliglustat twice daily | 171 | 8 y f/u results published 2018 Lukina | NCT00358150 | |||
| P2 | Untreated (for the past 12 m) GD1 patients aged 18-65 years, GBA1 enzyme deficiency, a spleen volume ten times that of normal, thrombocytopenia, and/or anemia. No splenectomy. | 28 | open-label, single-arm, 50mg bid | 52w | Statistically significant improvements in hemoglobin level, platelet count, spleen volume, liver volume, lumbar spine BMD, and biomarkers. | NCT00358150 | |
| P2 extension | An extension (one further year) of the above study | 20 | Improvements maintained and extended through the 4 years. | ||||
| P1 SAD | 0.01 to 30 mg/kg | ||||||
| P1 MAD | 50, 200, or 350 mg bid | ||||||
| P1 Food effect |
Ambroxol
Mechanism
| field | transcription |
|---|---|
| compound / row | Ambroxol / Mechanism |
| binding site | ABX bind to amino acid segments 243-249, 310-312, and 386-400 near the active site of GCase. |
| ER folding effect | stabilizing the native conformation of the mutant GCase in the ER, (이미 Misfolded된 것 피는게 아니라, 결합 후 misfolding을 예방함) |
| mechanism chain | ↑ normal folding & more functional molecules to form -> evade the ER-associated degradation pathway by UPS -> The protein-chaperone complex is safely transported to the lysosome -> the inhibitors will be displaced allowing GBA to hydrolyze substrate(18). |
| pH note | ideal PC for any lysosomal storage disease (LSD) should bind maximally at the neutral pH of the ER and minimally at the acidic pH of lysosomes. |
| question | Compound bind both mutated and WT? GBA Heteroz mutation 환자는 50개 정상 gba protein, 30개 mutated gba protein 을 생산할 것 (20정도는 ER에서 걸려서 파괴될테니) |
| active-site binding note |
What's the mechanism of "active site binding"? 답: Isofagomine binds to the active site of the β-glucocerebrosidase enzyme as it is folding. This was thought to help β-glucocerebrosidase fold correctly. https://gaucherdiseasenews.com/chaperone-therapy/isofagomine/ |
| 단점 | Null mutation에는 소용없음. (ie 한 allele은 null mutation, 다른 allele은 WT인 Heterozygous m) |
Preclinical Of Ambroxol
| row | GBA heterozygous model | PD only model | Normal mice | NHP |
|---|---|---|---|---|
| model | L444P/1 mice (Migdalska-Richards et al. 2016, PMID 27859541) | SNCA/SNCA mice (Migdalska-Richards et al. 2016, PMID 27859541) | (Luan, 2013 #1219) | |
| Tx | Ambroxol 12 days, L444P/1 mice | Ambroxol 12 days | ||
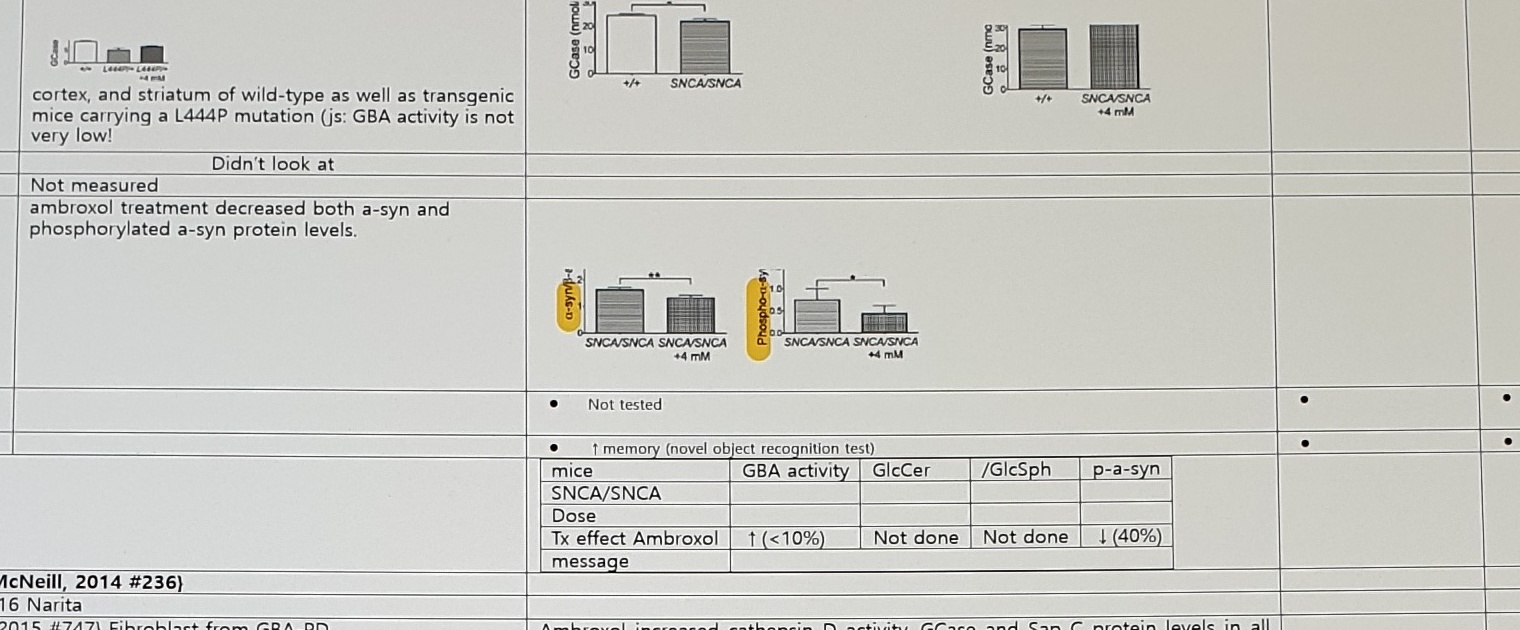
| GBA activity | increased GCase activity in the brainstem, midbrain, cortex, and striatum of wild-type as well as transgenic mice carrying a L444P mutation (js: GBA activity is not very low!) | Increased gba activity in brain, spleen, heart | ↑ GBA level | |
| GlcCER | Didn't look at | |||
| GlcSph | Not measured | |||
| a-syn / p-a-syn | ambroxol treatment decreased both a-syn and phosphorylated a-syn protein levels. | SNCA/SNCA mice; Tx effect Ambroxol: GBA activity ↑ (<10%), GlcCer Not done, /GlcSph Not done, p-a-syn ↓(40%). | ||
| Neuronal loss | Not tested | |||
| Behavior | ↑ memory (novel object recognition test) |
| source row | transcription |
|---|---|
| In vitro (McNeill, 2014 #236) | |
| Ex vivo 2016 Narita | |
| (Ambrosi, 2015 #747) Fibroblast from GBA-PD | Ambroxol increased cathepsin D activity, GCase and Sap C protein levels in all groups, while LIMP-2 levels were increased only in GBA1-mutant PD fibroblast. |
| (Luan, 2013 #1219) Fibroblast from GD | Ambroxol -> ↑ N370S, F213I, N188S/G193W & R120W mutant β-Glu activities. |
Clinical Trials Of Ambroxol
| population / sponsor | key eligibility / condition | status | pt # / ID | design | Tx | primary outcome | secondary / result notes |
|---|---|---|---|---|---|---|---|
| GD1 | suboptimal response to ERT | Ongoing | 60; NCT03950050 | Single group, OL | 12m | Platelet count, BMD, GlcSph | |
| PDD, Lawson Health Research Institute, Univer of Western Ontario | H&Y 2-3.5, mild to mod dementia (MMSE ≥16), MoCA ≤24, (No GBA genotyping) | Ongoing | 75; NCT02914366 | Ambroxol vs placebo, DB, randomized | 52w | ADAS-cog |
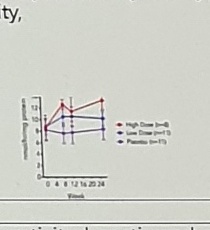
UPDRS III, MCI, CDR, Trail Making test, MMSE, PD-CRS; CSF biomarkers levels of αSyn (pg/ml), Tau (pg/ml), phospho-Tau (pg/ml) and β amyloid-42; MRI: brain ventricle volume (cm3) and hippocampal atrophy; GCase in lymphocytes; neurometabolites (MRS). [RESULTS AD/PD 2021] Dose-dependent (WBC) ↑ gba activity. |
| GD type 1, Exsar Corporation | Type 1 GD GBA deficiency resulting from mutations in the GBA genes, which have been shown to respond to Ambroxol in vitro screening assay | Suspended | 20; NCT01463215 | Single group, OL | 2m | safety | GBA activity, hepatic and splenic volumes from imaging scans, Lab (acid phosphatase, angiotensin-converting enzyme, serum bilirubin, Hb, plt, WBC, serum iron, clotting time, etc) |
| nGD | Ambroxol+ERT, nGD exhibiting significant chaperone effects confirmed by an in vitro test with patient-derived cultured skin fibroblasts (because chaperones, including ambroxol, are not always effective on all mutations) | 5 (3 adults, 1 child, 1 adolescent), 4 of them had prior ERT Tx. | Single group, OL | (Narita, 2016 #560) | Safety; target ambroxol dose (25 mg/kg/day or a maximum dose of 1300 mg/day) |
adult patients also okay (NARITA). GlcSph normalization not required (Narita). post-Tx GBA activity may remain low (AMC). baseline GlcSph may be high (narita). Dose-determination note: GCase activity in GD fibroblasts significantly enhanced with ambroxol >0.3-1 μM; previous study trough plasma concentration reached 1.7 μM at 1000 mg/day. | |
| nGD | Ambroxol+ERT. Narita patients had higher baseline GBA activity, while AMC patients all had approximately 5% at baseline. Conclusion note: we need patients with baseline GBA activity not very low. | 4 (3 adol, 1 adult), all of them had prior ERT Tx.; mean mSST score at baseline: 12.3 | Single group, OL | AMC, (Kim, 2020 #809) | Serum ambroxol exposure / dose-response note |
Only when Serum ambroxol exposure became > Narita (ie 27 mg/kg/day, after 2.5y), residual GBA activity (leukocyte) 5.1 -> 13.7%, GlcSph become normalized (in leukocyte/dried blood spots), reduced by 83% (dried blood spot) (45.7 -> 7.7 ng/mL), 4.8이 ref 이니 이는 거의 normalized된 것임. Sz, cognition, speech, pyramidal & extrapyramidal symptoms, mSST improved, BUT horizontal & vertical saccade no change, no change in brain MRS, unclear change in standing & walking balance. | |
| OUS; Italy |
NCT05287503; EudraCT 2021-... Inclusion: Duration of motor symptoms >5 years. Exclusion: PDD (MDS Level II criteria). | 52-week treatment period | Primary: i) MoCA change, ii) Rate of conversion from normal cognitive status to MCI or from MCI to overt dementia over the 52-week treatment period. |
Uncertain Spans
| location | unresolved text | reason |
|---|---|---|
| top carry-over row | first bullet outcome line in P3 EDGE row | The row begins above the visible crop edge and is only partially present here. |
| eliglustat top row | published 2018 Lukina | Citation note is very small but visually consistent with Lukina. |
| mechanism row | 386-400 | Numeric amino-acid segment is readable, but treated as high-risk numeric transcription. |
| mechanism question | 50개 정상 gba protein, 30개 mutated gba protein | Low-resolution Korean note; visually readable but kept as high-risk. |
| plot panels | exact axis values and labels | Embedded as image assets rather than guessed from OCR. |
| OUS Italy row | EudraCT 2021-... and right-side cells | The right portion of this row is clipped at the photo’s edge. |