aSyn - Vaccine — NPT200-11 row tail (Neuropore), Anle138b / Ionis / PD1601 row tail, AC Immune PD01A (ACI-7104) and PD03A AFFiRiS row group (P2 VacSYn / NCT06015841, P1 MSA / NCT02270489, P1 NCT01568099 / sPD), AFF008 study series, CSF Oligo-aSyn / MDS-UPDRS III, SAD study MEDI1341 PK data
NPT200-11 / aS aggregation inhibitor (Neuropore, NCT02606682)
P1
(NCT02606682)
preclinical
completed in 2016 by Neuropore
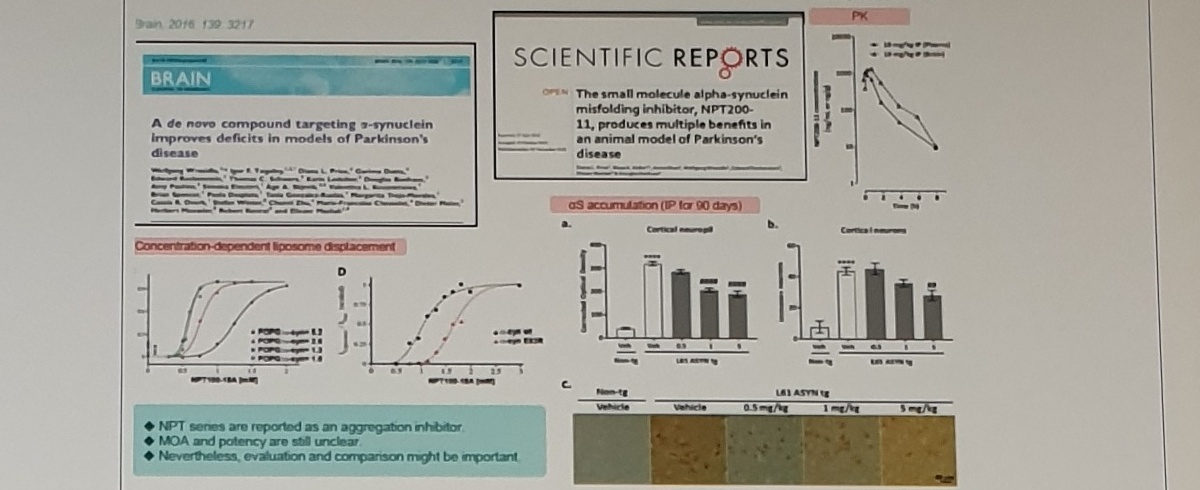
aS aggregation inhibitor NPT200-11
Scientific Reports
The small molecule alpha-synuclein misfolding inhibitor, NPT200-11, produces multiple benefits in an animal model of Parkinson’s disease.
aS accumulation (IP for 90 days)
NPT series are reported as an aggregation inhibitor. MOA and potency are still unclear. Nevertheless, evaluation and comparison might be important.
| item | company/source | modality/type | description | phase/status | note |
|---|---|---|---|---|---|
Anle138b | MODAG/TEVA | SM | specifically binds toxic oligomeric structures of aSyn, AND dissolves toxic oligomers | P1b (FIH) / completed | |
Ionis | against misfolded a-syn | the ip administration in non-transgenic mice intracerebrally injected with α-syn fibrils reduced LB pathology, decreased dopaminergic neuron loss, and overcame motor deficits (Tran et al., 2014) | |||
PD1601, PD1602 | BioArctic/Abbvie | Preclinical, No updates for this quarter report |
AC Immune: PD01A (ACI-7104) and PD03A AFFiRiS
AC Immune: PD01A (ACI-7104) and PD03A AFFiRiS are 2 novel therapeutic vaccine candidates
active immunotherapy
moa: induce α-syn aggregate-specific antibodies
a Specific Active Immunotherapy (SAIT), consisting of short antigenic peptides with amino acid sequences that mimic but not identical to the native α-syn species and has been shown to interfere with α-syn pathogenic mechanisms.11-13
This involves immunization with short peptides (AFFITOPEs) mimicking the aa sequence of a segment of the target protein.12 The respective AFFITOPE is conjugated to the carrier protein keyhole limpet hemocyanin (KLH) and is absorbed to aluminum hydroxide. The carrier protein provides the required T-helper epitopes for the induction of a longlasting and boostable antibody response, whereas the antigenic component (ie, the AFFITOPE) operates solely as a B-cell epitope and is responsible for the specificity of the humoral immune response.
P2 VacSYn / NCT06015841
an adaptive, biomarker-based Phase 2 study
The planned Phase 2 study is expected to have an adaptive design and it will be a two-part study:
Part 1 will evaluate Safety, Tolerability & Immunogenicity. Will include key immunogenicity measures and measures of pathological alpha-syn and alpha-syn aggregation, e.g. phospho-alpha-syn and alpha-syn-oligomers
Part 2 will be a proof-of-concept in early PD: Will include evaluation of motor and non-motor functioning (UPDRS based), neurodegeneration of dopaminergic terminals (DaT SPECT or VMAT2 imaging), digital biomarkers of motor and non-motor function, advanced MRI (including ASL and DTI), and functional and patient reported outcomes
Patients will enter Part 1 of the study and will be gradually transitioned to Part 2; All participants from Part 1 will contribute to the final analysis
Interim analysis is planned with biomarker data
Early immunogenicity data to inform dose and dosing schedule
AC Immune also believes that the interim analysis will aid to “understand biological signal for early transition to filing”
AFFiRiS and AC Immune are combining their expertise to develop new biomarkers to detect biological signals such as alpha-syn PET and fluid biomarkers
AC Immune believes that they might be able to use their alpha-syn PET tracer to select patients and follow the progression of the disease in this Phase 2 study; Data from AC Immune’s alpha-syn PET tracer study is expected in Q3 2021
P1, MSA (ClinicalTrial.gov identifier: NCT02270489)
P1, MSA (ClinicalTrial.gov identifier: NCT02270489).
(Meissner, #905) FIH (this was also presented at MDS 2020) randomized to receive 5 subcutaneous injections of either PD01A (n = 12), PD03A (n = 12), or placebo (n = 6) in this patient- and examiner-blinded, placebo controlled, 52-week phase 1 clinical trial
[Immunological Results:]
89% of treated patients developed a PD01-specific antibody response after receiving all injections.
Induced antibodies displayed clear reactivity to the α-syn target epitope.
Titers and antibody responder rate (58%) were lower in the PD03A treated group.
O (MDS2020) PD01A induced a long-lasting humoral immune response in PD patients that can be reactivated upon booster application
Conclusion: PD01A triggered a rapid and long-lasting antibody response that specifically targeted the α-syn epitope.
P1, NCT01568099 (sPD)
P1, NCT01568099
sPD,
no evidence for genetic forms of the disease
Early PD (≤ 4 years), H&Y I/II
Age between 45 and 65 years or age between 40 and 45 years if there is no evidence for genetic forms of the disease
(Volc, 2020 #932) a randomized, controlled, parallel-group, patient-blinded, and single-center pilot study, but open!
3.5년 f/u!
Immunological [ Time Frame: 12 month ]
- Titer of vaccination induced antibodies directed towards vaccine components, alpha- and β syn
Clinical Activity [ Time Frame: 12 month ]
- Change in motor symptoms (MDS-UPDRS III)
- Change in non-motor PD symptoms (e.g.; MDS-UPDRS Ia, II, PDQ39, cognition)
- Change in biological and radiological markers (e.g. CSF alpha syn levels)
[Immunological Results:]
O (MDS2020) PD01A induced a long-lasting humoral immune response in PD patients that can be reactivated upon booster application
O Human PD01A-induced antibodies specifically and selectively bind aggregated aSyn species
O SPR analysis of week 26 CSF and serum samples from three high responding patients revealed that the concentration of PD01A elicited antibodies in CSF relative to serum was about 0.3%
AFF008 study series
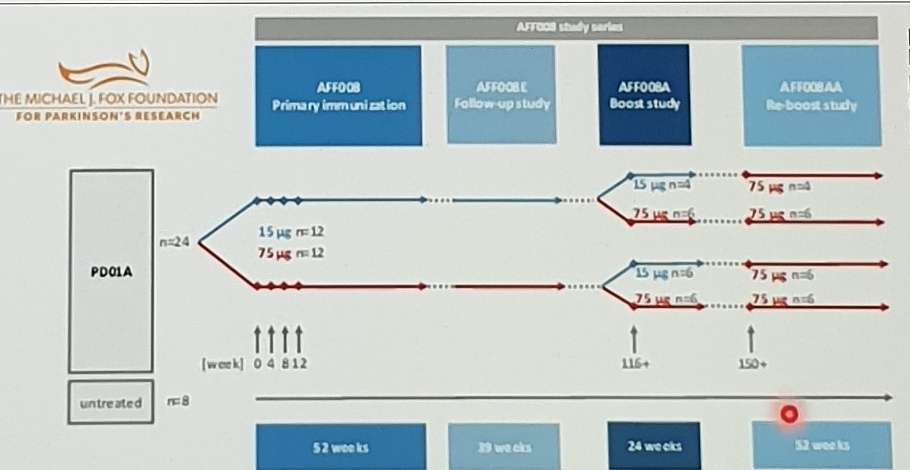
[Clinical Results:]
Monitoring of patient’s safety during the Phase 1 study series over 3.5 years did not show any safety concerns associated with PD01A administration
MDS-UPDRS I, II, III scores in treated groups showed no or only minor increase over the study period
(위 그림엔 untreated group 있으나, volc 논문엔 없음. 어차피 open 임. 그래서 MDS-UPDRS score 점수비교 없음)
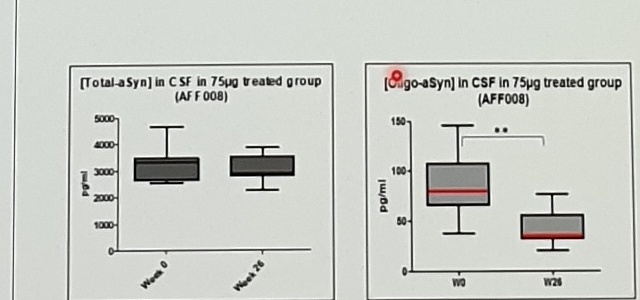
CSF Oligo-aSyn concentrations correlated with MDS-UPDRS III
High dose PD01 treatment was associated with a 50% Lowering of Pathological aSyn (Oligo-aSyn) in CSF
CSF Oligo-aSyn / MDS-UPDRS III
[↓ CSF Oligomeric aSyn]
CSF samples, collected at screening and at week 26
The concentration of oligomeric α-syn in the CSF was analysed post hoc using ELISA as described by Majbour and colleagues;23 the lower limit of this assay was reported to be 10 pg/mL. Briefly, the conformation-specific mouse monoclonal antibody Syn-O2 was used for capturing α-syn oligomers. Detection was done using the rabbit polyclonal antibody FL-140 (Santa Cruz Biotechnology, Santa Cruz, CA, USA), followed by the second step antibody goat anti-rabbit IgG labelled with HRP (Jackson ImmunoResearch, West Grove, PA, USA). Specific binding was visualised using an enhanced chemiluminescence substrate (SuperSignal ELISA Femto, Pierce Biotechnology, Rockford, IL, USA) and measured by an Envision plate reader (PerkinElmer, Waltham, MA, USA).
Early reduction of CSF Oligo-aSyn correlated with longitudinal changes of MDS-UPDRS III scores
lxiii) PD01A-induced antibody titers correlated with the stabilization of MDS-UPDRS III scores over the course of the study
lxiv) Treatment with the 15 μg dose also led to a slight decrease in the oligomeric α-syn (appendix p 5).
lxv) Further analyses revealed a significant correlation (Pearson correlation coefficient r=0.515 and p=0.020) between the oligo-α-syn concentration in the CSF at baseline and baseline MDS-UPDRS part 3 scores
- DAT-SPECT examinations did not show statistically significant changes up to 91 weeks in study 1 (appendix p 4).
- MDS-UPDRS part 3 scores were generally stable across the studies.
- Mean MDS-UPDRS part 3 scores in the pooled 15 μg group were 11.9 (SD 8.2) at baseline and 12.5 (14.8) at the last
SAD study - PK data to date
AstraZeneca / Takeda
MEDI1341 exposure
| Parameter | 70 mg | 210 mg | 400 mg | 1200 mg |
|---|---|---|---|---|
| Dose increment | - | 3.00 | 1.90 | 3.00 |
| Increase in Cmax | - | 3.75 | 1.51 | 3.17 |
| DPF Cmax | - | 1.25 | 0.79 | 1.06 |
| Increase in AUC0-∞ | - | 3.44 | 2.04 | 2.58 |
| DPF AUC0-∞ | - | 1.15 | 1.07 | 0.86 |
At the 1200-mg dose level, CSF/serum MEDI1341 concentration ratios were assessed on Days 8 and 15;
Uncertain Spans
| location | transcription | uncertainty |
|---|---|---|
| prior table row alignment | source/phase/status association for some rows | row boundaries visible, but some cells are partly cut by crop/scroll position. |