PARKIN GT Decision Tree, Draft Timeline, Biomarker Matrix
Clinical Trial Endpoint Continuation
Stable fragments:
4. Novel approach. Ph1/2 may require a large-scale clinical trial design and probably endpoints are indeterminate.
Even for Ph1/2, it may impossible to find trial patients in a reasonable time frame.
- Prkn only with placebo (? N=12 to 14)
- DBS only, with placebo (? N=6 or use natural Hx data)
- Prkn + DBS, with placebo (? N=12)
Endpoint:
DBS symptomatic but Prkn disease modifying: so different endpoint timingDecision Flow Notes
Highlighted note before the figure:
Decision Flow
아래 부분은 좀 그림과 안 맞으니, 최종 그림이 나오면 다시 정리하자
[Xin]
- good results from GBA in May2022
- NHP studies with both ICM and neurosurgery -> determine ROA (2023/03)
[Takeshi]
- May 2022: ROA ICM vs neurosurgery for 4 AAV capsidsPARKIN GT Decision Tree
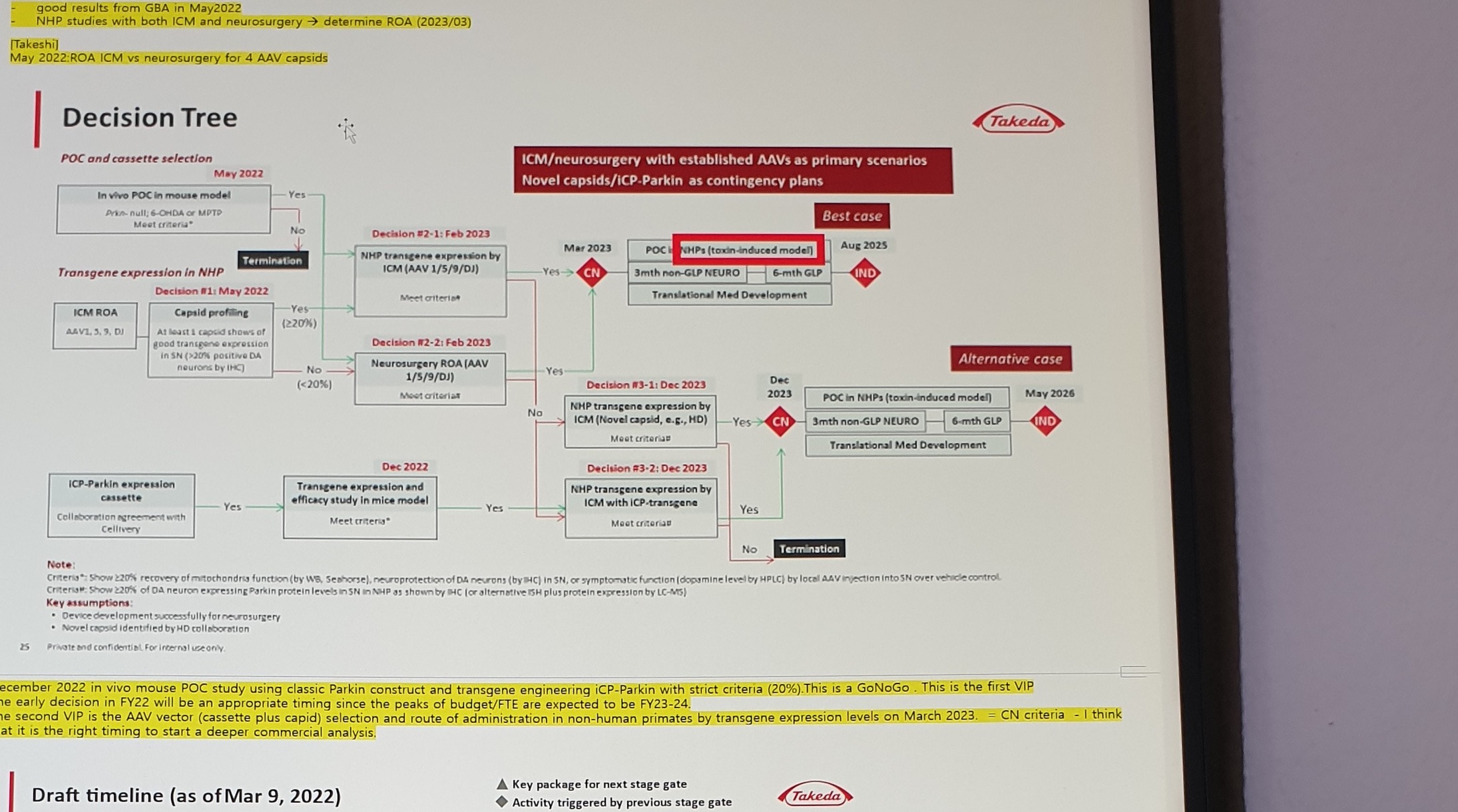
Stable figure text fragments:
Decision Tree
POC and cassette selection
ICM/neurosurgery with established AAVs as primary scenarios
Novel capsids/iCP-Parkin as contingency plansDecision / activity labels:
| Area | Stable text |
|---|---|
| Mouse POC | In vivo POC in mouse model; Prkn-null; 6-OHDA or MPTP; Meet criteria*; May 2022; Termination |
| NHP expression | Transgene expression in NHP; Decision #1: May 2022; ICM ROA; AAV1, 5, 9, DJ; Capsid profiling; At least 1 capsid shows of good transgene expression in SN (>20% positive DA neurons by IHC) |
| Established AAV path | Decision #2-1: Feb 2023; NHP transgene expression by ICM (AAV 1/5/9/DJ); Decision #2-2: Feb 2023; Neurosurgery ROA (AAV 1/5/9/DJ) |
| iCP-Parkin path | ICP-Parkin expression cassette; Collaboration agreement with Cellivery; Dec 2022; Transgene expression and efficacy study in mice model |
| Best case | Mar 2023; CN; POC in NHPs (toxin-induced model); 3mth non-GLP NEURO; 6-mth GLP; Translational Med Development; Aug 2025; IND |
| Alternative case | Decision #3-1: Dec 2023; NHP transgene expression by ICM (Novel capsid, e.g., HD); Decision #3-2: Dec 2023; NHP transgene expression by ICM with iCP-transgene; Dec 2023; CN; May 2026; IND |
Figure notes:
Criteria*: Show >=20% recovery of mitochondria function (by WB, Seahorse), neuroprotection of DA neurons (by IHC) in SN, or symptomatic function (dopamine level by HPLC) by local AAV injection into SN over vehicle control.
Criteria#: Show >=20% of DA neuron expressing Parkin protein levels in SN in NHP as shown by IHC (or alternative ISH plus protein expression by LC-MS).
Key assumptions:
- Device development successfully for neurosurgery
- Novel capsid identified by HD collaborationHighlighted bottom note:
December 2022 in vivo mouse POC study using classic Parkin construct and transgene engineering iCP-Parkin with strict criteria (20%). This is a GoNoGo. This is the first VIP.
The early decision in FY22 will be an appropriate timing since the peaks of budget/FTE are expected to be FY23-24.
The second VIP is the AAV vector (cassette plus capsid) selection and route of administration in non-human primates by transgene expression levels on March 2023. = CN criteria - I think that it is the right timing to start a deeper commercial analysis.Draft Timeline
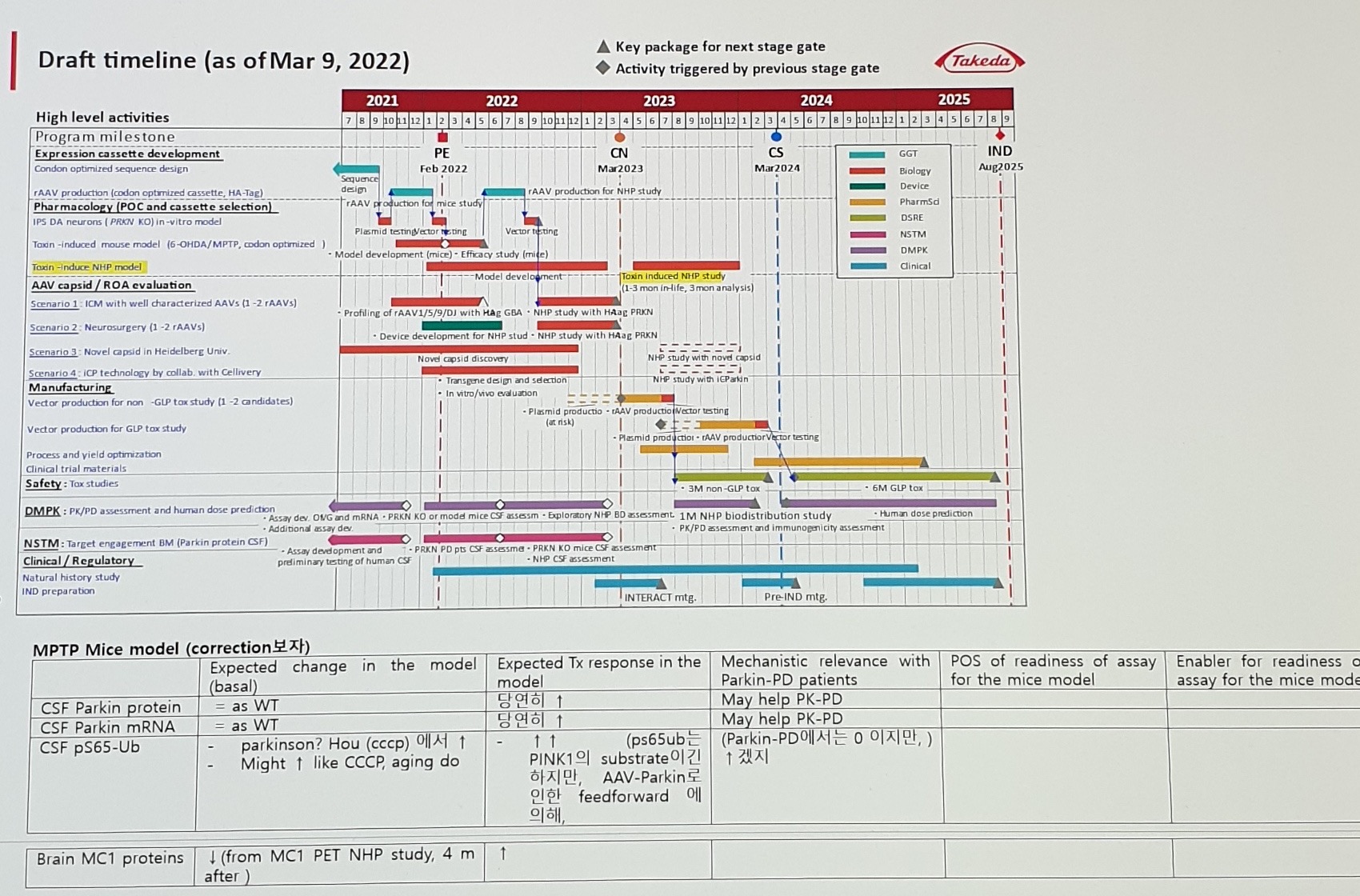
Header:
Draft timeline (as of Mar 9, 2022)
Key package for next stage gate
Activity triggered by previous stage gateMilestone labels:
| Gate / milestone | Stable text |
|---|---|
PE | Feb 2022 |
CN | Mar 2023 |
CS | Mar 2024 |
IND | Aug 2025 |
Legend categories:
GGT
Biology
Device
PharmSd
DSRE
NSTM
DMPK
ClinicalHigh-level activity rows:
Program milestone
Expression cassette development
- Codon optimized sequence design
- rAAV production (codon optimized cassette, HA-Tag)
Pharmacology (POC and cassette selection)
- IPS DA neurons (PRKN KO) in-vitro model
- Toxin-induced mouse model (6-OHDA/MPTP, codon optimized)
- Toxin-induce NHP model
AAV capsid / ROA evaluation
- Scenario 1: ICM with well characterized AAVs (1-2 rAAVs)
- Scenario 2: Neurosurgery (1-2 rAAVs)
- Scenario 3: Novel capsid in Heidelberg Univ.
- Scenario 4: iCP technology by collab. with Cellivery
Manufacturing
- Vector production for non-GLP tox study (1-2 candidates)
- Vector production for GLP tox study
- Process and yield optimization
- Clinical trial materials
Safety: Tox studies
DMPK: PK/PD assessment and human dose prediction
NSTM: Target engagement BM (Parkin protein CSF)
Clinical / Regulatory
- Natural history study
- IND preparationAdditional activity fragments:
Toxin induced NHP study (1-3 mon in-life, 3 mon analysis)
NHP study with HAag PRKN
NHP study with novel capsid
NHP study with iCParkin
3M non-GLP tox
6M GLP tox
Exploratory NHP BD assessment
1M NHP biodistribution study
PK/PD assessment and immunogenicity assessment
Human dose prediction
Assay dev. OVG and mRNA
PRKN KO or model mice CSF assessment
PRKN PD pts CSF assessment
NHP CSF assessment
INTERACT mtg.
Pre-IND mtg.Biomarker Expected-Change Matrices
MPTP Mice Model
Header:
MPTP Mice model (correction보자)Columns:
Expected change in the model (basal)
Expected Tx response in the model
Mechanistic relevance with Parkin-PD patients
POS of readiness of assay for the mice model
Enabler for readiness of assay for the mice modelRows:
| Biomarker | Expected change in the model (basal) | Expected Tx response in the model | Mechanistic relevance with Parkin-PD patients |
|---|---|---|---|
CSF Parkin protein | = as WT | 당연히 ↑ | May help PK-PD |
CSF Parkin mRNA | = as WT | 당연히 ↑ | May help PK-PD |
CSF pS65-Ub | parkinson? Hou (cccp) 에서 ↑; Might ↑ like CCCP, aging do | ↑↑; Korean note about PINK1 substrate and AAV-Parkin feedforward | (Parkin-PD에서는 0 이지만,) ↑겠지 |
Brain MC1 proteins | ↓ (from MC1 PET NHP study, 4 m after) | ↑ | blank / not safely readable |
Parkin KO Mice Model
Rows:
| Biomarker | Expected change in the model (basal) | Expected Tx response in the model | Mechanistic relevance with Parkin-PD patients |
|---|---|---|---|
CSF Parkin protein | none | 당연히 ↑ | May help PK-PD |
CSF Parkin mRNA | None | 당연히 ↑ | May help PK-PD |
CSF pS65-Ub | Very low (from postmortem papers) | ↑ | May help PK-PD |
Brain MC1 proteins | ↓ (from Palacino) | ↑ | May help PK-PD |
PRC1 In-Vivo Disease-Relevant / Function Model Dataset
Title and subheaders:
Candidates Of In Vivo Disease-relevant/Function Model For PRC1 Dataset (Draft)
Model establishment in Parkin KO mice compared to WT mice (-Oct 10, 2021)
Efficacy study of rAAV9-Parkin in Parkin KO mice (-Oct 29, 2021)Data criteria:
| Section | Minimum data | Preferable / Best / Nice-to-have data |
|---|---|---|
| Model establishment in Parkin KO mice compared to WT mice | Reduction of Parkin activity (e.g. phospho-poly ubiquitination) | Reduction in mitophagy; Increase in mitochondrial protein (Tom 20); Decrease in autophagy levels (LC3-II); best data: Reduction in number of DA neurons (e.g. TH cell or TH fiber) |
| Efficacy study of rAAV9-Parkin in Parkin KO mice | Significantly increase in Parkin activity | best data: Significantly reverse mitophagy deficits; nice to have: Reverse reduction of TH |
The left model-description column appears to include:
6-OHDA model or
Ethanol model or
Spontaneous model (young adult)Publication And IP Notes
The bottom table appears under a partially cut heading, likely Finalization.
Rows/fragments:
| Item | Publication | IP |
|---|---|---|
PRKN protein / brain parkin assay | Publication planned for brain parkin assay (collaboration with Juntendo-U, publication timing TBD). IP는 안 할거지만 publication 은 해서 so that we can protect the assay originality from competitors | Takeda does not care about IP for assay method because it's not our core business |
pS65Ub | Will; IP는 안 할거지만 publication 은 해서 so that we can protect the assay originality from competitors | X (since we've got help from Mitokinin, it's not good to get IP by ourselves.) |
Uncertain Spans
- Decision tree figure labels and arrow logic are image-primary; use the embedded decision-tree asset before extracting exact gate logic.
- Draft timeline month-level bars and dense activity labels are image-primary; use the embedded timeline asset before extracting exact schedule fields.