iCP-Parkin, USP30/NHP Plan, MitoCoP, And DBS+PRkn Strategy
GBA PET Rationale Carry-Over
비록, (Efficacy 없이) PK-PD relationship 관점만 보면 (가장 가능성 있는 PD index인)
CSF GlcCer 와 가장 잘 맞을 것은 CSF (total) GBA protein 혹은
whole brain (total) GBA PET signal 이겠지만,
- 나중에 aSyn PET 과 regional pattern 맞춰봐야함),
[부가적 rationale:]
CSF GBA protein
- No healthy data (이건 향후 보유 예정)
- No consensus on CSF GBA BL level in PD (only Mullin 2020 Ambroxol P2 data),
that shows variability 150-310 pg/mol
CSF GBA ACTIVITY
- (CSF니까) Affected by lysosomal- & cellular- secretion
- Variable: adult normal range: 1.1-8.1 umol/L/d; prevail.
- In some patients, undetectable (LLOQ; 0.56 umol/L/d).GBA Expression Evaluation By Imaging
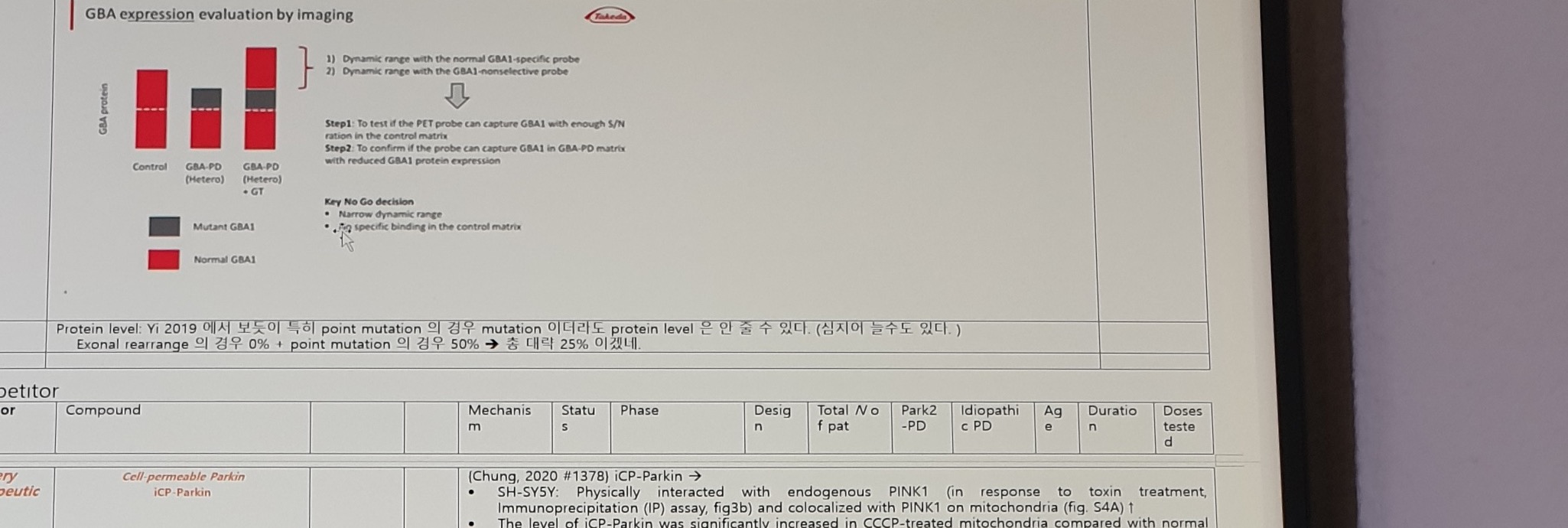
Figure text:
GBA expression evaluation by imaging
1) Dynamic range with the normal GBA1-specific probe
2) Dynamic range with the GBA1-nonselective probe
Step1: To test if the PET probe can capture GBA1 with enough S/N ratio in the control matrix
Step2: To confirm if the probe can capture GBA1 in GBA-PD matrix with reduced GBA1 protein expression
Key No Go decision
- Narrow dynamic range
- Non-specific binding in the control matrixLegend/labels:
| Label | Text |
|---|---|
| Y-axis | GBA protein |
| Groups | Control; GBA-PD (Hetero); GBA-PD (Hetero) + GT |
| Legend | Mutant GBA1; Normal GBA1 |
Korean note:
Protein level: Yi 2019 에서 보듯이 특히 point mutation 의 경우 mutation 이더라도,
protein level 은 안 줄 수 있다. (심지어 늘수도 있다.)
Exonal rearrange 의 경우 0% + point mutation 의 경우 50% -> 총 대략 25% 이겠네.Competitor Table: iCP-Parkin And USP30/NHP Plan
Competitor table header columns include Sponsor, Compound, Mechanism, Status, Phase, Design, Total N of pat, Park2-PD, Idiopathic PD, Age, Duration, and Doses tested.
Cellivery Therapeutics / iCP-Parkin
| Field | Content |
|---|---|
| Sponsor | Cellivery Therapeutics |
| Compound | Cell-permeable Parkin; iCP-Parkin |
| Evidence anchor | (Chung, 2020 #1378) iCP-Parkin -> |
In-vitro / mechanism notes:
SH-SY5Y: Physically interacted with endogenous PINK1 (in response to toxin treatment,
Immunoprecipitation (IP) assay, fig3b) and colocalized with PINK1 on mitochondria (fig. S4A) ↑
The level of iCP-Parkin was significantly increased in CCCP-treated mitochondria compared
with normal mitochondria (FIG3C, WB)
(ELISA). The amount of iCP-Parkin was significantly increased in the CCCP-treated
(in vitro cells) PARKIN KO cells
↑ ubiquitination of Miro2 (fig3e), PARIS
↑ mitophagy (confocal microscope, fig3g)
↑ mitochondrial proteins: cytochrome c oxidase I (COX1), succinate dehydrogenase
complex flavoprotein subunit A (SDH-A; Fig. 3H), Tom20, translocase of the inner
membrane 23 (Tim23), MFN1 and MFN2
↑ expression of genes involved in mitochondrial biogenesis: peroxisome proliferator-activated
receptor gamma coactivator 1? (PGC-1?), transcription factor A, mitochondrial (TFAM),
and nuclear respiratory factor 1 and 2 (NRF1 and NRF2; Fig. 3I and fig. S4I).Correction notes:
[correction-in vitro]
CCCP and MPP induced dose-dependent [increase?] in ROS, reduced ATP levels,
and increased apoptosis that were recovered by iCP-Parkin
(Fig. 3, J to L, and fig. S4, J and K) in a dose-dependent manner (fig. S4L)
In SH-SY5Y engineered to overexpress aSyn:
Oligomeric and filamentous alpha-syn were significantly decreased by 93 and 80%
in the soluble fraction (Fig. 4B).
iCP-Parkin significantly reduced pSer129-alpha-syn, and total aSyn.
Cf) iCP-Parkin physically interacted with pSer129-alpha-syn and synphilin-1
[correction-in vivo]
6OHDA mice: restored TH loss (WB, fig5d), restored (fig. S6D) COX4, VDAC1
6OHDA mice: restored TH loss (fig5j) and plasma DA levels (fig5k)
MPTP mice: restored urine DA levels (fig. S7C) and TH expression (fig. S7,F, G, and I).
AAV-aSyn: restored TH-positive neurons in SN (FIG6C), and striatum (fig s8b),
reduced aSyn level in TH+ neurons by 72 and 79% in SN and the striatum, respectively
(Fig. 6, D and E). Protein aggregates visualized by thioflavin S staining were also
significantly reduced (76%) in SN (Fig. 6J), also similarly observed in the striatum
(fig. S8D), and reduced the levels of pathological [filamentous/aggregated
(Fig. 6K and fig. S8H) and phosphorylated (Fig. 6, L and M)] aSyn and GFAP
(fig. S8G) in the striatum and/or SN. Behavior test fig6, rotarod, pole test.USP30 Inhibitor / MTX115325 / NHP Plan
| Row / field | Content |
|---|---|
| Sponsor | MISSION Therapeutic; NysnoBio |
| Compound | USP30 inhibitor; brain-penetrant USP30 inhibitor, MTX115325; https://www.nysnobio.com/pipelin[e] |
| Modality | Small molecule; later AAV5; later ASO |
| Scientific advisor | Nobutaka Hattori (scientific advisory board) |
| Preclinical anchor | (Fang, 2023 #2558) i) USP30 KO + AAV A53T aSyn mouse; ii) MTX115325 + AAV A53T aSyn mouse |
| PRKN KO rat | 1.0e11 total vg; 26 w; Unilateral |
| PRKN KO mouse | ↓ IL-6 (ELISA); PRKN-PD: ↑ IL-6 (Papers and Nysnobio data) |
NHP (Macaca fascicularis) (BD) | AAV5; 1.4e12 total vg; 6w; Unilateral -> IHC; Korean note: 잘나왔다고 사진; Neuronal and glial Parkin transduction with sparse coverage of SN DA neurons; quantification X |
NHP (safety) | 1.4e12 total vg; 6w; 3.6e12 total vg; 18w |
| Clinic / plan | Apple Watch |
| Resource | 20221220 project team meeting |
MitoCoP/ASO row:
Mitophagy Compensator for PARK2-PD patients (MitoCoP)
ie mitophagy inducer
ASO
NEXT: In vitro POC (neuroprotection) by knockdown of 40 candidate hit-genes
via siRNA introduction to PARK2-null iPS-DAn
-> nomination of the most promising target for ASO development.DBS+PRkn Strategy
Bottom content of the DBS+PRkn strategy table is partially cut off by the photo boundary.
| Item | Left/claim | Pros | Cons / notes |
|---|---|---|---|
| Intro | might be technically feasible to administer | ||
1 | Treatment timelines are different (not interchangeable) | DBS patients are older, PD has progressed and DBS is for symptomatic treatment (motor fluctuations, dyskinesia, tremors resistant to medication). Prkn patients are younger; genetically identified, non-symptomatic | |
- | Treatment targets are different (not interchangeable) | - | DBS anatomical targets are STN, GP, possibly PN, while Prkn may be P, SN, SN+P. |
2 | Primarily we want to treat cause not effect; expected treatment benefits likely not interchangeable | Prkn will treat young/early onset expecting Prkn benefit. Prkn patients have no motor symptoms to treat by DBS. Therefore no DBS benefit to justify DBS related risks. IF treat late/progressed Prkn patients with DBS, then Prkn benefit will be low to zero. No Prkn benefit to justify Prkn related risks | |
3 | Dual treatment doubles several risks | Expands the number of targets in brain that have an intervention (catheter, catheter + electrode). Increases surgery risks, invasiveness, limits future DBS treatment if Prkn fails and vice versa Adds the risk from DBS implanted component failure and revision surger[y] | |
4 | Novel approach. Ph1/2 may require a large-scale clinical trial design and probably endpoints are indeterminate | Even for Ph1/2, it may impossible to find trial patients in a reasonable time frame. - Prkn only with placebo (? N=12 to 14) - DBS only, with placebo (? N=6 or use natural Hx data) - Prkn + DBS, with placebo (? N=12) | |
Endpoint: | DBS symptomatic but Prkn disease modifying; so different endpoint timing | ||
5. |
Uncertain Spans
- Top GBA block likely belongs to a preceding GBA PET rationale section; it should not be treated as a new Parkin/PARKN GT subsection without adjacent-page confirmation.
CCCP and MPP induced dose-dependent [increase?] in ROSis reconstructed from context/OCR and remains uncertain.