Parkin Biomarker Strategy And Fibroblast Mitochondrial Evidence
Ubiquitin Linkage Basics
Top text:
Ubiquitination occurs through a linkage between the C-terminus of ubiquitin and the e-amino group of a lysine residue on the target substrate.
Ubiquitin itself has seven Lys residues (K6, K11, K27, K29, K33, K48, and K63).
Ubiquitin to form polymers through various lysineThe final sentence appears cropped/truncated at the right edge.
Biomarker Discovery And Validation Questions
Misa san,
- BM discovery 할건기?
- Biosample sources: NCNP, J-PPMI 에서 CSF, Fibroblast, PBMC 얻을 수 있나?
- Other candidates도 있는데: MFN (Can be measured in human PBMC), VDAC1
- BM validation & qualification
- mouse에서 할 건가?
- 한다면 어느 tissue? Brain parenchyme도 볼건가?
- Human sample에서?
- Fibroblast, PBMC studies 가능한기?
- KOL 어떻게 만날수 있나? Natural Hx study of GBA+PDD
- Dr Narita at Tottori university for GD consultation.pS65-Ub, Parkin Substrates, TE Biomarker, And Patient Selection
- pS65 ub
- this would be a good candidate pharmacodynamic BM, because exogenous Parkin significantly increases this in vitro (Fiesel, 2015), reflecting enhanced formation of poly-Ub chains.
- This is likely low in Park2-mutant PD, because Parkin deficiency was associated with low level of pS65 ub in vitro (2018 Ordureau fig 6, in vitro) (2018 Sliter), but Do we need to confirm this in postmortem PD Brain of Park2-mutant PD (to assess disease relevance)?
- OTHER biomarkers of pharmacodynamic?
- Parkin substrate?
- Eg. VDAC1, which is an abundant mitochondrial outer membrane protein
- Any other bm of mitophagy (more directly reflecting the functional level of mitophagy?
- Biomarker of TE?
- PARK2 mRNA and Parkin protein? Need to set up the assay of these?
- NDE?
- Patient selection
- Park2 mutations in parkin-mutant PD are variable (although a few mutation, eg p.R275W, is more commonly found than other mutations). Could we do in-vitro screening in patients in a clinical trial of AAV-Parkin before we actually confirm including a patient? i.e. In a patient-derived fibroblast culture, measure the level of mitophagy to confirm they have Loss of function mutation (ie mitophagy is not induced by CCCP) and whether it is improved by functional WT Parkin (plasmid PARKIN or AAV-PARKIN)?
- Examples
- Ambroxol Narita, Exsar
- Disease-related bm
- This will be dopaminergic neuronal loss (histology in preclinical and DATscan in clinic)Fibroblast Mitochondrial Function Rationale
Fibroblast
- Function
- to secrete extracellular matrix, which forms the connective tissue of an organ
- Mitochondria
- Fission, complex I, basal mitochondrial membrane potential (MMP) (ψm)
- (Antony, 2020 #713) similarities in mitochondrial dysfunction between fibroblasts and neurons
- Key question: (compensatory morphological increase도 흔한데,) can MC1 density represent MC activity?MC1 Density, MC1 Activity, And De-Risking Strategy
| Context | MC1 density | MC1 activity | Notes / challenge |
|---|---|---|---|
| Human | Mortiboys: not sufficiently support (이것도 MC1 specific X); Pacelli: swelling; Van der Merwe: swelling; Grunewald: ↑ mito mass; 결국, 1. MC1 만 본 것 없음. 2. Overlap in Mortiboys, ↑ mito mass in Grunewald | Brain: 영원히 모르므로, Fibroblast_충분히 support중 (Mortiboys, Pacelli), 반증은 Grunewald 2010 | Schapira 1990: MC1 density & activity are not paired.; Keeney, 2006 #944) 에서는 Correlation 되는 듯 (with 34% decrease in its 8 kDa subunit & ↓ MC1 activity) |
| animal | Noda: number는 증가라고 suppose!; Stevens: mt number & size는 꽤 overlap됨.; MtDNA COPY number로는 꽤 차이; 결국, MC1 만 본 것 없음. | No evidence (TBD) | Aim: Calibration -> Tx response, but 한참후에야 실험 가능. |
De-risking strategy table anchors:
| Strategy | Proposed assay / evidence | Challenge |
|---|---|---|
| In vitro | Parkin-PD fibroblast, MC1 density & activity | Sample: Juntento univ, Niigata univ; MC1 density 볼수있나 confirm? (CLeeter가 porin봤으나, MC1만 본 건 못 찾겠는데?) |
| In vivo | mostly blank | Availability of aged-mice (not by Nov 30, 2021) |
| MJFF funding | row label |
Respiratory Complex Stoichiometry And Function Summary
C1-selective deficit 원인: 2016 GRUNEWALD:
The stoichiometric ratios of the respiratory chain complexes have been measured as CI, 0.8-1.3; CII, 1.2-1.4; CIII, 3; and CIV, 6.0-7.5 in bovine heart mitochondria. Thus, CIV may exist in excess, whereas CI levels may be limiting (C1 적으니 잘 준다?).
Moreover, the quantification of respiratory chain protein turnover rates in mice showed that the subunits of CII have the shortest average half-life, followed by the components of CI and CIV, whereas the components of CIII have the longest halflife. (CIII 는 오래 가니 안 준다?)Summary before the small table:
For MC1:
Mortiboys, gen O, ↓ mc1 activity,
Zanellati, gene O, ↓ ATP, ↓ MMP
van der Merwe, gene O, ↓ ATP, swelling,Small summary table:
| Study/model | Number | Area (density) | activity | ATP |
|---|---|---|---|---|
| Mortiboys | blank | ? (rather ↑ mito branching) | ↓ | ↓ |
| KO mice (Noda) | ↑ | ↓ | ? | ? |
Pacelli 2011 excerpt below the table:
(2011 Pacelli) The decreased mitochondrial ATP synthesis by CI- and CII-substrates, observed in patients' fibroblasts, is compensated by an increase in the anaerobic glycolytic pathway. A general shift to anaerobic glycolysis in the neocortex of PD patients has been established by MRS and 2-[18F]fluoro-2-deoxy-D-glucose PET, showing increased lactate concentrations [60].Skin Fibroblast General Notes And Mortiboys/Zanellati Evidence
General notes:
| Area | Skin fibroblast - general / Parkin mutant PD |
|---|---|
| gene | Express most of the PARK genes at relevant levels including PINK1 and Parkin genes; SNCA gene is barely expressed; the vesicle/receptor/ion channel control is rather rudimentary in fibroblasts |
| cell in general | Reflect cumulative cell damage at the age of the patient |
| mitochondria function | Dynamic is a little different; MMP 잴 수 있음 (dye -> confocal microscopy using fluorescent light -> Image J) (Au - Burbulla, 2012 #717); Mito morphology 볼 수 있음 (eg. fragmented network) (Au - Burbulla, 2012 #717); Mitophagy 볼수 있음 (colocalization with lysosome using mitotracker & lysotracker -> Image J) (Au - Burbulla, 2012 #717) |
Mortiboys patient mutation table anchors:
| Patient | First mutation | Second mutation |
|---|---|---|
| Patient 1 | 255del_A het (exon 2) | Deletion exon 5 |
| Patient 2 | 202_203delAG (exon 2) | 202_203delAG (exon 2) |
| Patient 3 | 202_203delAGhet (exon 2) | Deletion exon 2 |
| Patient 4 | 202_203delAG (exon 2) | Deletion exon 4 |
| Patient 5 | c.101-102delAG; p.Gln34ArgfsX38 | c.1289G>A; p.Gly430Asp |
Mortiboys note:
(Mortiboys, 2008 #719) n=5, genotyping available (table1) (vs health control)
(patients 42 +/- 5.8 years, 개별나이는 없음), no individual patient ages shown.
- ↓ MMP (fig1, by 45%) MC1 activity and ↓ (fig2a, by 48%, complex I linked ATP-production), ↓ (fig2c, by 58%) ATP level, ↑ mito branching, small decrease (20%) in Mitochondrial number per cell (without statistical significance, fig3d)
- =MC II activity, =(FIG2b, MC3-linked ATP production),
- Why MC1 specific? : no comment
- KD of parkin (↓ by 80%) in control fibroblasts -> ↓ MMP, ↓ ATP levels, ↑ mito branching
- but, 50% kd of parkin, did not result in impaired mitochondrial function or morphologyZanellati note:
(Zanellati, 2015 #712) n=4, protein level data 있음. (vs health control)
↓ MMP (fig3, ~half), ↑ OCR (consistent, in all mutant samples, compensatory Fig2A, seahorse), ↓ ATP (fig2c, ~half, pt 간 나이차에도 불구하고 atp 차이 별로 없음, 오히려 14세가 최고 감소), = mito morphology ('network' number of cells with tubular & fragmented network, measured by MitoTracker, 의미: damaged mito 가 fission 되지 그대로 network 에 남아 악영향주고있군)Pacelli, van der Merwe, Grunewald, And Zilocchi Evidence
Pacelli note:
(Pacelli, 2011 #725) n=2 (P1, 36 years old, and P2, 32 years old) no genotyping (vs health control) (Takanori slide 에는 delEx2-3/delEx3)
mito structural abnormality (swelling), ↓ (by 32%, no overlap) MC1 (table1), IV activities, =complex II activity, ↓ citrate synthase activity, impaired energy metabolism ROS, lipid oxidation, peroxisomal alteration, but ↑ ATP, ↑ (extracellular) lactate, (↑ meaning compensatory glycolytic pathway) PGC-1α was remarkably upregulated in both patients. However, the function of PGC-1α was blocked, as revealed by the lack of its downstream target gene induction
Why MC1 specific? : no commentvan der Merwe patient table anchors:
| Patient lab ID | Gender & ethnicity | AAO (yr) | Age in 2013 (yr) | Length of disease (yr) | Parkin mutation | Zygosity |
|---|---|---|---|---|---|---|
P1 (53.44) | F; MA | 27 | 37 | 10 | Del Ex3-4 | Homozygous |
P2 (56.45) | F; W | 27 | 50 | 23 | Del Ex4 | Homozygous |
P3 (56.43) | F; W | 27 | 52 | 25 | Del Ex4 | Homozygous |
van der Merwe note:
(van der Merwe, 2014 #722) n=3, P2 is 13 years older than P1 (but 둘이 ATP 감소 정도 유사함, variable 해서 설명난해), (vs health control)
protein level 그림없네. South Africa, ↓ ATP (fig 3), =morphology (branching), swollen mitochondria, ↑ intracellular electron-dense vacuoles (js: aggregated damaged mt might be possible),Grunewald note:
Negative study 이네! (Grünewald, 2010 #724) 개별 환자, clinical data 는 (Pramstaller, 2005 #2043)에 존재. genotyping data 유 (table1). protein level 자료안보임. n=6, age: 41-79, ↓ ATP (fig1, both synthesis (↓30%) & concentration (↓60%), 연령과 비교 불가함), but =MC1 activity (p3 좌본문), = (13% reduction) functional MC1 content (NADH ferricyanide reductase assay, which allows to determine the content of functional complex I, 원문과 달리 이건 function 인 듯), ↑ mito mass (measured by citrate synthase activity), MMP 는 유사, network branching basal was similar, but more decrease under paraquat, ↑ Oxidized proteins, =branchingZilocchi note:
(Zilocchi, 2020 #901) n=5, 개별 환자자료유, genotyping data 있음. Age: 15-75. Proteomics, ↓ MMP (fig2c, dmso상황에서 이미 낮고 CCCP로 추가 감소 없음. 나이 다양한데도 별로 variance 없이 감소되어 있음.), = mito network morphology (authors: While DRP1 localizes to depolarized mitochondria, OPA1 and MFN1 levels are not altered. As a whole, it seems that even if fission is triggered (DRP1 accumulation), fusion is not blocked (lack of OPA1 and MFN1 elimination). No PINK1 accumulation (아예 not detectible at baseline) CCCP treatment determined the accumulation of PINK1 in control subjects. On the other hand, PINK1 levels were drastically reduced in skin fibroblasts from PARK2-mutated patients exposed to CCCP,Zilocchi Patient Table And Parkin Western Blot Figure
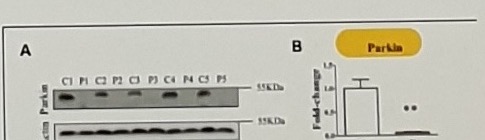
Title and figure caption anchors:
TABLE 1 | Outline of primary skin fibroblast cells from PARK2-mutated Parkinson's disease patients and control subjects.
FIGURE 1 | Parkin levels are reduced in PARK2-mutated patients.
(A) Representative western blot image of Parkin protein in primary skin fibroblasts of PARK2-mutated patients (P1, P2, P3, P4, and P5) and control subjects (C1, C2, C3, C4, and C5). (B) Relative quantification expressed as ...Patient table anchors:
| Subject | Age at onset | Age at skin biopsy | PARK2 mutations |
|---|---|---|---|
| P1 | 60-65 | 70-75 | Del_1/p.R275W |
| P2 | 15-20 | 40-45 | Del_3-4-5/p.R33X |
| P3 | 10-15 | 15-20 | Dup_2/Del_3-4-5 |
| P4 | 40-45 | 50-55 | p.Q34Rfs x X5 homo |
| P5 | 20-25 | 30-35 | p.Thr240Met/Del3 |
| C1 | CTRL | 25-30 | CTRL |
| C2 | CTRL | 25-30 | CTRL |
| C3 | CTRL | 30-35 | CTRL |
Uncertain Spans
BM discovery 할건기?/BM validation & qualification: OCR and image agree on meaning, but the exact Korean colloquial ending may be할건가?.J-PPMI,NCNP,MFN,VDAC1,PBMC, andCSFappear in the source text but may need normalization if used as structured metadata.Juntento univmay beJuntendo univ; the photo text appears closer toJuntento/Juntendo, so the source spelling is retained.CLeeterin the MC1 density challenge is likely a person/reference shorthand, but the spelling is unclear.MC1may refer to mitochondrial complex I (MCI) in many places; the original page consistently showsMC1or local shorthand, so it is preserved.- The direction arrows in Mortiboys, Zanellati, Pacelli, van der Merwe, Grunewald, and Zilocchi notes are small and uncertain.
- Mutation strings are small and uncertain:
p.R275W,c.1289G>A,p.Gly430Asp,c.714C>G,c.823C>T,p.Q34Rfs,p.R33X,p.Thr240Met/Del3,Del_3-4-5, and related exon labels. p.Q34Rfs x X5 homoin the Zilocchi patient table is visually difficult.- Zanellati patient-table
Code,Age of onset,DNA, andProteinentries are partially visible and uncertain. - The Zilocchi figure caption is cut off at the bottom of the photo.
- Page label
Page 107 of 113repeats across nearby photos, so this is probably a scroll/capture position within the same Word page rather than a unique source page.