Parkin Substrates, Animal Models, And Parkin-PD Prevalence
Substrates And Functions Of Parkin
| substrate / label | function / notes | pathway / category |
|---|---|---|
VDAC1 / preceding row fragment | VDAC1 is one of the most strongly ubiquitylated PARKIN substrates (Ordureau, 2018 #698); Quantification of VDAC1 ubiquitination 가능하다는 것 같은데 ? (is this Fig 4G ?) (Ordureau, 2018 #698); caveat notes conflicting reports, with evidence both for (Geisler et al., 2010) and against (Narendra et al., 2010a) VDAC involvement in PARKIN-dependent mitophagy. | |
PARIS (ZNF746) | 75, 76, 181, 182, 190, 191]; mitochondrial biogenesis by ubiquitination of PARIS (PGC-1_[Peroxisome proliferator-activated receptor gamma coactivator-1-alpha] transcriptional repressor, Lee et al., 2017) | |
Miro | mitochondrial movement by regulating Miro (Wang et al., 2011; Gaweda-Walerych and Zekanowski, 2013) | |
aminoacyl tRNA synthetase complex interacting multifunctional protein 2 (AIMP2), | ||
synphilin-1 ( | an alpha-syn interacting protein that is enriched in the core of Lewy bodies and ubiquitinated by parkin in a manner abolished by familial PD-associated mutations. | |
CDC-rel1, cyclin E, , Pael-R, XI, sp22 | ||
Mitofusins Mfn1 and Mfn2, | which are large GTPases that promote mitochondria fusion into dynamic, tubular complexes that maximise efficiency of oxidative phosphorylation | |
Additionally, Parkin contains a C terminal motif that binds PDZ domains.the p38 subunit of aminoacyl-tRNA synthetase complex[36] and far upstream element-binding protein 1[37] through addition of Lys48-linked poly-Ub chains | Parkin has been shown to associate in a PDZ dependent manner with the PDZ domain containing proteins CASK and PICK1 | UPS |
Tubulin | Axonal transport | |
synaptotagmin XI (SYT11) | Synaptic Vesicle fusion | |
P53 | parkin protects dopaminergic neurons from cytotoxicity induced by PD-mimetic 6-OHDA, mediated by suppression of neuronal p53 expression and its downstream activation of the apoptotic cascade. Several PD-associated parkin mutations are localised to RING1 and might impair the ability to bind and downregulate the p53 promoter, leading to enhanced p53 expression. Parkin-mutant PD patients also exhibit a four-fold elevation in p53 immunoreactivity. | |
parkin itself | ||
aSyn (?) | Parkin, has been previously demonstrated to protect against the toxic effects of alpha-syn, https://www.michaeljfox.org/grant/investigating-effect-parkin-alpha-syn-propagation |
UPS
Parkin also enhances cell survival by suppressing both mitochondria dependent and -independent apoptosis.Parkin PD - Prevalence Opening Note
Parkin PD
Prevalence(Poorkaj, 2004 #572) 18% of familial PD (js 전체 PD의 1.8%네), However, if parkin is recessive, only 5% of early-onset cases who had compound mutations could be attributed to this locus. Mutation frequency was 0.12 (95% CI 0.04-0.19). 70% of parkin cases were heterozygous. It is unclear whether heterozygous mutations are pathogenicAnimal Models Of Parkin PD
Animal models of Parkin PD
overexpression of T240R-parkin and Human WT parkin
progressive and dose-dependent DA cell death [146].
Parkin-Q311X-DAT-BAC mice exhibit multiple late onsets and progressive| model / citation | background or model detail | mitochondrial abnormality | neurodegeneration / phenotype | Lewy body / note |
|---|---|---|---|---|
PARKIN KO mice(Noda, 2020 #701) | B6;129 genetic background; Only At the age of 110 weeks, locomotor impairments, including hindlimb defects, began to show motor behavioral deficits at 110 weeks | [SN 만 봄]; Only At the age of 110 weeks mitochondrial fragmentation in DA neuron in SN, small (↓ (fig2b, by 60%) mt area) fragmented mt accumulate (↑ the number of mt per unit of cytosolic area) in both cytosol & outside cells, ↓ mt area; EM: fragmented mitochondria had normal outer membrane structure (Fig. 3A c inset), but displayed irregular inner structure, i.e., the structures of matrix and cristae were broken. | Only at 120 w old; TH+ neurons: ↓ (30%?) in SNcc, = in VTA; DA, DOPAC, HVA: ↓ (20%?) in dorsal striatum; young mice had very little phenotypes other than downregulation of DA release and upregulation of D1 and D2 dopamine receptors in the striatum; TH neurons not decreased in 48-week-old mice (원문에는 3 month 인데?) (Sato et al., 2006); 12 m: DA, DOPAC, HVA 도 Striatum 에서 안 줌 (Sato 2006); Js: 1 년째에서 별 변화 없어서 (Sato 2006) 2 년째 봤구나 (Noda 2020) | No LBs |
(Palacino, 2004 #726) | [ventral midbrain including SN]; [8 m old]: ↓ MC1 proteins (24kDa subunit, -28% 아마 NDUFV2) & (30 kDa subunit, -9%, 아마 NDUFS3), ↓ (반) Cytochrome c oxidase protein (subunit Vb); continued fragment: ↓ MC1 (by 17%), 2 (by 25%), 3&4 (by 23%)-linked ATP generation; But, normal morphology | decreased abundance of a number of proteins involved in mitochondrial function or oxidative stress. Consistent with reductions in several subunits of complexes I and IV, functional assays showed reductions in respiratory capacity of striatal mitochondria isolated from parkin/ mice. Electron microscopic analysis revealed no gross morphological abnormalities in striatal mitochondria of parkin/ mice. | ||
(Stevens, 2015 #1031) | conditional KO of Parkin in the adult mice ventral midbrain (lentivirus expressing GFPCre) | ↓ (32% but overlap) mitochondria number and ↓ (40% but overlap) size, ↓ mtDNA COPy number (fig 2, ~half?) | dopaminergic neuronal loss | |
(Pinto, 2018 #934)PD-mito-PstI mouse | 원래 In this model: mitochondrial DNA undergoes double-strand breaks in the dopaminergic neurons only, (ie acquired insult (mtDNA damage) that has been observed in normal aging.); increase in mtDNA levels (mutant and wild type).; exhibit a small but significant decrease in the number of mitochondria in the dopaminergic axons only; it did not cause a parallel increase in mitochondrial mass or mitophagy.; Our results suggest that Parkin affects mtDNA levels in a mitophagy-independent manner.; we observed a small but significant decrease in mitochondrial number only in the dopaminergic axons of ParkinKO-PD-mito-PstI mito-eYFP mice (Fig. 6C). When we analyzed mitochondrial size, we observed that the lack of Parkin led to the accumulation of larger mitochondria (Fig. 6D,E). We also examined the steady-state levels of the following two proteins associated with mitochondrial dynamics: Mfn2 and OPA1, which is involved in the fusion of the outer and the inner mitochondrial membrane, respectively. Although we did not detect changes in Mfn2 levels, there was a slight decrease in OPA1 (longform) levels in striatal homogenates from 4- and 12-month-old animals (Fig. 6B and Fig. 4-1J available at https://doi.org/10.1523/JNEUROSCI.1384-17.2017.f4-1). | |||
(Perez and Palmiter, 2005). | B6;129 genetic background, | Perez and Palmiter claimed no loss of neurons at ages up to 22 months (88 weeks) | ||
(Itier et al., 2003). | ||||
(Goldberg et al., 2003), | ||||
(Von Coelln et al., 2004), | ||||
Parkin KO rat / Horizon, TGRL4760 | Neurobiol Dis. 2014;70:190-203 | |||
Parkin KO crossed with mutator KI mice (Dr. Youle at NIH) | (Pickrell, 2015 #702) Neuron. 2015;87(2):371-381 | |||
Parkin-Q311X Tg mice (Jax, 009090) | (Lu, 2009 #930) J Neurosci. 2009;29(7):1962-1976 | Dopaminergic Neuron Degeneration, late-onset and progressive hypokinetic motor deficits | Expressing a Truncated Mutant Parkin, Accumulation of Proteinase K-Resistant aSyn | |
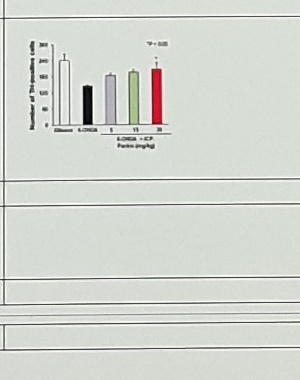
6-OHDA Rat | (Vercammen, 2006 #703) | Correction: Lentivirus-Parkin + | ||
6-OHDA mice | Sci Adv. 2020;6(18):eaba1193 | |||
P mice | (Bian, 2012 #704) | Correction: Parkin Tg + | ||
2-Parkin/rat aSyn expression | (Yamada, 2005 #705) |
Prevalence Of Parkin Mutations
| group | text |
|---|---|
| familial | familial |
| sporadic | sporadic |
Juvenile Parkinson's (JP)
young onset PD (21-40)
recent consensus seems
% of total PDFamilial
20% of YOPD patients have at least one first- or second-degree relative with PD either in the same or antecedent generation. (Klepac, 2013 #690)
Parkin mutation: PARKIN pathogenic mutation has been found in 50% cases of familial PD and up to 15% of early onset sporadic cases in one cohort (Lücking, 2000 #693) although a recent meta-analysis (Kilarski, 2012 #692) concluded that the figure is around 15.5% of familial and 4.3% of sporadic cases.
아래로 정리 20200924 tDIB:
Kilarski 2012: 136 명의 EOPD cases 중 29 가 familial cases, so 21%
Kilarski 2012, 100 x 10% x 21% x 15.5% (the prevalence of PARK2 mutations in patients, p1525 에 3952 patients 중에서의 prevalence 라고 명시했고, patient 니까 homoz 혹은 compound heteroz 이겠지)
= 0.33% of total PD = in the us, 3,255 casesSporadic
Majority cases of YOPD (80% 잡으면), 다른 reference?
- PARK2 mutation: 15% of YOPD (<45)).
Then, 100 x 10% x 80% x 15% = 1.2% of total PD
But according to Kilarski 2012, 100 x 10% (early onset) x 79% x 4.3% (the prevalence of PARK2 mutations in patients, p1525 에 3952 patients 중에서의 prevalence 라고 명시함, patient 니까 homoz 혹은 compound heteroz 이겠지)
= 0.34% of total PD = in the us, 3,397 cases
(Chaudhary, 2006 #928) YOPD (N=173)중 2 명만이 HOMOz or compound hetero
아래로 정리 20200924 tDIB:
- 79% of YOPD cases are sporadic
(Kilarski et al. 2012, doi 10.1002/mds.25132)
- 4.3% of sporadic YOPD have PARK2 mutations
(Kilarski et al. 2012, doi 10.1002/mds.25132)
=> 0.34% of total PD (~3,000 cases in the US)Combined Young-Onset Parkin-PD Estimate
Young onset Parkin-PD (Bi allelic) is 0.67% (0.33+0.34) in total PD. So 0.67% of total PD cases worldwide (6 million) is 402,000 cases.
(Alcalay, 2010 #691) 16.6% of YOPD patients (without regard to family history of PD.) have mutations (including (6.7%) PRKN, (3.6%) LRRK2, (6.7%) GBA, and (0.2%) DJ1 -> then Parkin-mutant PD is 3.6% of YOPD without regard to family history of PD. (ie 0.36% of total PD cases)Uncertain Spans
| location | text/status | reason |
|---|---|---|
| VDAC1 row | Fig 4G ?, Ordureau, 2018 #698, conflicting reports for/against VDAC involvement | Top of row is cut off and citations are small. |
| Noda model | TH+ neurons ↓ (30%?), DA/DOPAC/HVA ↓ (20%?), SNcc, 48-week-old vs Korean note 3 month 인데? | Numeric percentages and age interpretation are explicitly uncertain in the source note. |
| Stevens model | ↓ (32% but overlap), ↓ (40% but overlap), mtDNA COPy number (fig 2, ~half?) | Values are visible but source note flags overlap/approximation. |
| 6-OHDA mice figure | bar plot labels and values | Figure is too small; asset preserved rather than transcribed. |